
Shining 3D — Aoralscan Elite & Aoralscan 3
Shining 3D is a Chinese 3D scanning company founded in 2004 whose dental division produces the Aoralscan family of intraoral scanners. Best known among scanner buyers for being one of the value-positioned alternatives to 3Shape and iTero, Shining 3D's flagship Aoralscan Elite was the first intraoral scanner with integrated photogrammetry (IPG) for full-arch implant accuracy. This page covers the two currently-positioned models — Aoralscan Elite and Aoralscan 3 — plus parent company, manufacturing, FDA status, and Shining 3D's own competitive claims.
About Shining 3D
Shining 3D is a publicly-traded Chinese 3D vision technology company. The company started in industrial metrology and reverse-engineering 3D scanners, then extended into digital dentistry with the Aoralscan family. Per Shining 3D's published company profile, they serve customers in 100+ countries with international sales representing more than 60% of revenue.
The company holds a substantial certification stack — ISO 9001, ISO 13485 (medical device quality), ISO 14001, ISO 45001, ISO/IEC 27001, ISO/IEC 27701, ISO/IEC 27017 (cloud security), ISO/IEC 27018 (PII protection), TISAX, Korea KGMP, MDSAP, CE, FDA, and FCC registrations. The breadth of these certifications is notable for a value-positioned scanner brand.
Product Timeline
- 2004Shining 3D Tech Co., Ltd. founded in Hangzhou as an industrial 3D-scanner company.
- 2018Aoralscan 1 launched, Shining 3D's first intraoral scanner — the dental brand begins commercial expansion outside China.
- 2020Shining 3D lists on the Shanghai Stock Exchange STAR Market, providing growth capital for the dental and metrology divisions.
- 2021Aoralscan 3 released — broader-adoption mid-range model that established Shining 3D in the global IOS market.
- 2023–2024Aoralscan Elite launched with integrated intraoral photogrammetry (IPG) — per Shining 3D, the first intraoral scanner with built-in photogrammetry for full-arch implant cases.
- 2026Aoralscan Elite receives an iF Design Award 2026; new Aoralscan Elf (entry-level) launched.
Current Intraoral Scanner Lineup (2026)
Shining 3D has the widest IOS lineup of any manufacturer covered on this site — a flagship (Aoralscan Elite, with optional Wireless), a proven mid-range (Aoralscan 3, with Wireless), a specialty unit (Aoralscan Lync — deep scan depth for denture & scan-body work), and a new entry-level tier (Aoralscan Elf). All four are covered below.

Aoralscan Elite (IPG)
Flagship · First IOS with intraoral photogrammetry
Shining 3D's current flagship — the first intraoral scanner with integrated intraoral photogrammetry (IPG), designed primarily for full-arch implant accuracy at conventional photogrammetry-system precision but without the extra device. Also sold as Aoralscan Elite Wireless IPG. iF Design Award 2026 winner.
Aoralscan 3
Proven mid-range workhorse
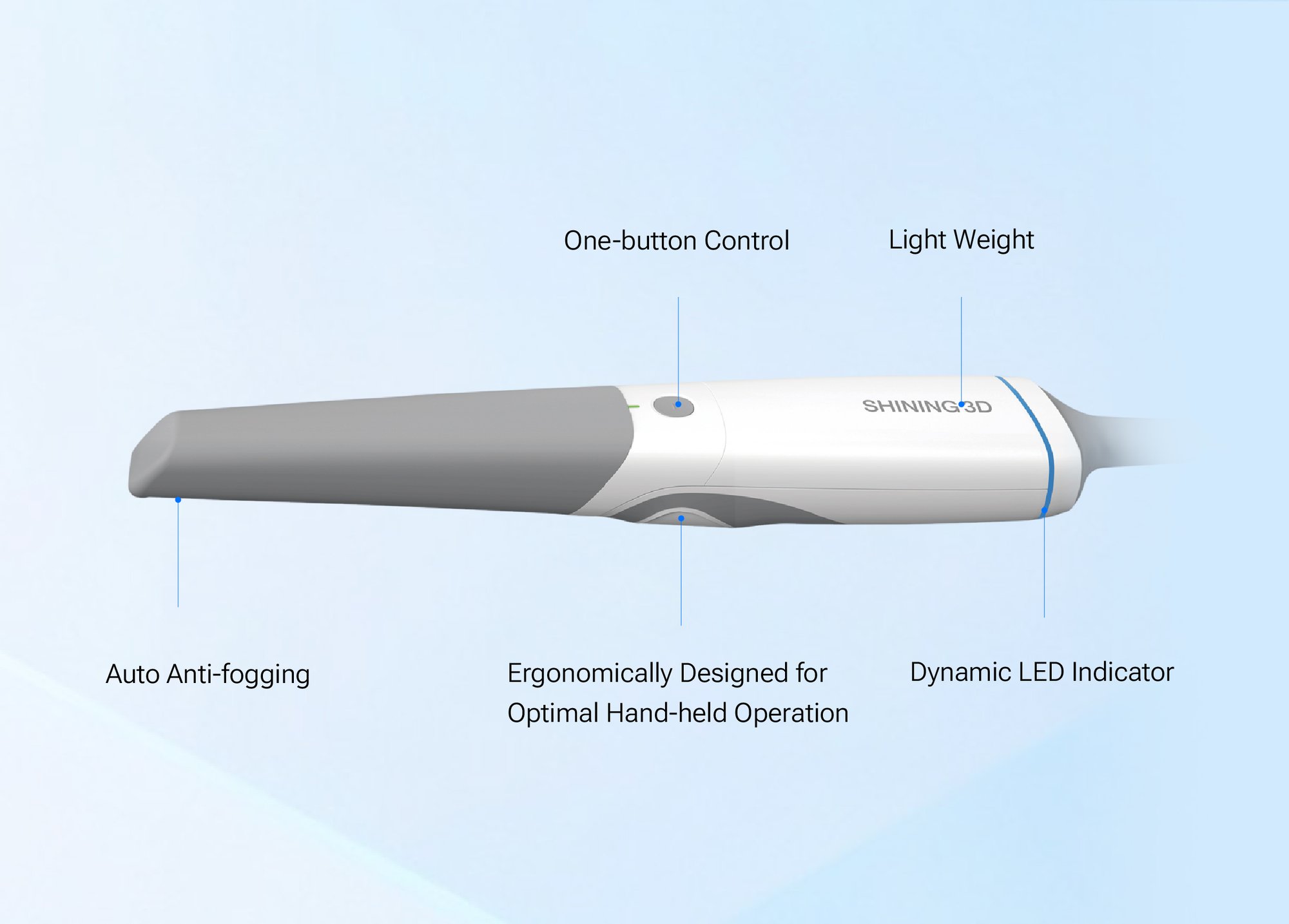
The model that established Shining 3D in the global IOS market — light-weight wand, AI-assisted scanning, motion-sensing wake/sleep, auto anti-fogging. Sold as a wired and wireless variant. Positioned by Shining 3D as a value-tier alternative to flagship-priced scanners.

Aoralscan Lync
Specialty · Deep scan depth (22 mm)
A specialty wand positioned by Shining 3D as "Less is More." 116 g, single Type-C plug-and-play cable, pen-like grip, and up to 22 mm scan depth — designed for denture impressions, scan-body capture, and periodontal scans where reach matters. Motion sensing + AI scan-speed-up are standard. Outputs open STL/OBJ/PLY.

Aoralscan Elf
New entry · "Smarter Occlusion"
Shining 3D's newest entry-tier scanner. Marketed as "Smarter Occlusion" — features IntelliBite AI-driven occlusal analysis, 3D plaque scanning & visualization for patient communication, and a Smart Report Comparison feature for tracking oral-hygiene outcomes between visits. Weighs just 106 g, with three interchangeable scanning tips and a single Type-C cable.
Product imagery: Shining 3D Tech Co., Ltd. Reproduced here for reference; please contact Shining 3D for licensing inquiries.
What is Intraoral Photogrammetry (IPG)?
Photogrammetry — using multiple synchronized cameras to triangulate the precise position of markers attached to implant scan bodies — is the gold-standard accuracy method for full-arch implant cases. Historically photogrammetry required a separate dedicated device (e.g., PIC dental, ICam4D) costing ~$30K–$50K on top of a regular intraoral scanner.
Aoralscan Elite IPG integrates a photogrammetry mode inside the intraoral scanner wand itself. Per Shining 3D, this allows full-arch implant cases to be captured with photogrammetry-grade accuracy from the same single device used for routine scans, without buying a dedicated photogrammetry system. Several published case studies on Shining 3D's site describe full-arch immediate-load workflows using IPG to verify implant positions before prosthetic design.
Source: shining3ddental.com — What is Intraoral Photogrammetry/IPG? · Independent third-party accuracy benchmarks are encouraged for any clinician relying on photogrammetry for high-precision full-arch cases.
What Shining 3D Claim as Competitive Advantages
The following are Shining 3D's own published positioning points. We list them as Shining 3D presents them — 3DL has not independently benchmarked these claims.
- Integrated intraoral photogrammetry (Aoralscan Elite). Per Shining 3D, the first IOS with built-in photogrammetry — eliminating the need for a separate ~$30K–$50K photogrammetry device for full-arch implant accuracy.
- 20+ years of 3D vision R&D. Shining 3D positions itself as a metrology-first company that brought industrial-precision know-how into dental scanning. Their corporate R&D team is 500+ engineers, ~40% of total workforce.
- Cost-effective alternative. Shining 3D positions Aoralscan 3 as a flagship-feature IOS at a value-tier price point versus the premium-tier 3Shape / iTero / Primescan flagships.
- AI-assisted scanning & auto features. Aoralscan 3 markets motion-sensing wake/sleep, auto anti-fogging, automatic scan clean-up, and AI-assisted scanning workflows.
- Open architecture / exports. Shining 3D scanners are positioned as exporting open STL/PLY without lab-routing lock-in to a single proprietary cloud — a workflow advantage for labs that maintain their own design pipelines.
- Complete digital workflow. Shining 3D Dental positions its end-to-end stack as Scan (Aoralscan) → Design (AI Design or Classic Design Service) → Print (AccuFab series) — one vendor for the full chairside-or-lab pipeline.
- Extensive medical certification. ISO 13485, MDSAP, KGMP, CE, FDA registration, ISO 27001/27701/27017/27018, TISAX — Shining 3D actively highlights this stack as evidence of regulatory and information-security maturity, particularly relevant for buyers concerned about the brand's Chinese parentage.
All claims sourced from Shining 3D's published product and corporate pages.
FDA & Regulatory Status
Shining 3D Tech Co., Ltd. holds FDA Establishment Registration #3010173003 as a foreign device manufacturer (China). The Aoralscan family is registered under FDA product code NOF ("System, Optical Impression, Computer Assisted Design and Manufacturing of Dental Restorations"), a Class II classification under regulation 872.3661.
| FDA Record | Type | Status |
|---|---|---|
Reg #3010173003 | Establishment Registration (foreign manufacturer) | Active, listed for NOF (optical impression) + NDP (implant accessories) + NFS (jaw tracking) |
Important nuance: Our openFDA search did not surface a specific 510(k) clearance under "Shining 3D" or "Aoralscan" device names. A device cleared under product code NOF is Class II — most NOF devices on the US market have an active 510(k) clearance held either by the manufacturer directly or by a US distributor. U.S. clinicians evaluating an Aoralscan purchase should ask Shining 3D's US distributor to provide the specific 510(k) number applicable to the configuration they're buying, and verify it in the FDA's public 510(k) database before committing.
Cloud Storage & Data Residency
Shining 3D's dental software stack writes scans to Shining 3D Dental Cloud, the company's case-routing and storage platform. Like most non-3Shape scanner vendors, Shining 3D does not publish a public region-by-region data-residency table.
Compliance Status
- ISO/IEC 27001 (Information Security Management) — certified at the company level.
- ISO/IEC 27701 (Privacy Information Management) — certified.
- ISO/IEC 27017 (Cloud-service-specific information security controls) — certified.
- ISO/IEC 27018 (Personally Identifiable Information protection in public clouds) — certified.
- TISAX (Trusted Information Security Assessment Exchange, automotive-grade standard widely used in EU industry) — certified.
- Data origin. Shining 3D's data-protection documentation indicates compliance with applicable Chinese, EU (GDPR), and US data-protection laws. The combination of TISAX + ISO 27018 is notable for a cloud platform.
Practical consideration for US dental practices: Shining 3D is a Chinese-parented company, which raises legitimate questions for some practices about cross-border data handling, China's Personal Information Protection Law / Data Security Law, and access by foreign regulators. Practices with strict patient-data-residency policies (e.g., DoD-affiliated, federal contractor) should request a written sub-processor and region disclosure from Shining 3D before signing — and verify whether a HIPAA Business Associate Agreement is available for U.S. PHI before uploading identifiable patient information to Shining 3D Dental Cloud. Shining 3D's certification stack is strong, but the cross-border angle is a separate operational question worth resolving in writing.
Working With 3DL on an Aoralscan Workflow
3DL accepts Aoralscan scans through two routes:
- STL / PLY / OBJ upload through the doctor portal at 3dlmi.com — Aoralscan exports open mesh formats by default, so cases come into our 3Shape Dental System design pipeline natively without a converter step.
- Shining 3D Dental Cloud invitation — share a case directly to a 3DL account from inside the Aoralscan software (works for both Elite and Aoralscan 3).
For full-arch implant cases captured with the Aoralscan Elite IPG mode, please call us before submitting so we can confirm the photogrammetry export format and verify scan-body library coverage for your specific implant system: (248) 919-7929.
Send Your First Aoralscan Case
Open a free account and submit a scan — your first single-unit crown is on us.